DMPK (drug metabolism and pharmacokinetics) studies are used for the rational selection of suitable drug candidates in the context of the development of a (bio)pharmaceutical drug. Due to many years of expertise, Axolabs can offer its customers DMPK activities in the following areas:
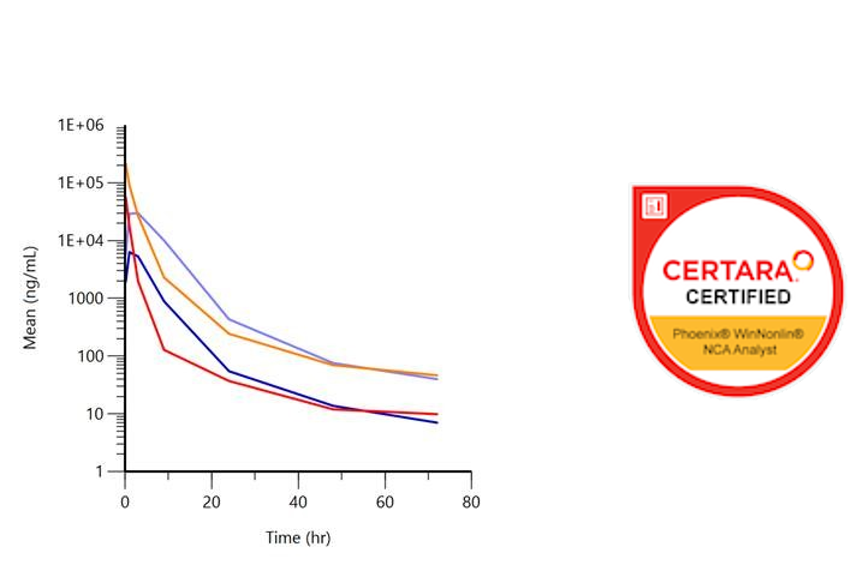
Evaluation of concentration-time data of preclinical and clinical studies (plasma and urine) determined in-house as NCA (Non-Compartmental Analyses) using the Phoenix WinNonlin® software. The analysis delivers meaningful graphs as well as important parameters for planning further studies, such as half-life, maximum exposure (cmax), time of maximum exposure (tmax), total exposure (AUC; area under the curve), volume of distribution (Vz) and clearance (Cl).